
Somatic Cancer
Liquid Biopsy
SHOP PRODUCTSAccuracy at extreme levels of sensitivity is crucial to the clinical utility of highly multiplexed NGS-based liquid biopsy assays. These tests demand purpose-built, patient-like reference materials to verify assay sensitivity and performance before delivering critical patient results.
SeraCare’s patent-pending circulating tumor DNA (ctDNA) technology ensures the most patient-like ctDNA sizing, clinically-relevant variant coverage, and improved library complexity to accelerate liquid biopsy NGS assay development, validation, and routine clinical implementation.
Comprehensive solutions across all phases of the NGS workflow
ctDNA v4
Our flagship ctDNA product containing 93 actionable variants and 10-fold less background noise compared to ctDNA v2 and ctDNA Complete. Available from 5% VAF to 0.1% in both purified DNA and plasma formats.
MRD now in single vials
You asked, we listened! Our MRD product is now available as individual tumor percentage vials in addition to the existing 4 vial kit.
ctDNA v2 & ctDNA CompleteTM
- Comprehensive ctDNA reference materials containing 40 clinically relevant pan-cancer variants (ctDNA v2) or 25 solid tumor variants including three CNVs (ctDNA Complete).
- Patient-like sized blend of contrived variant constructs and background wild-type DNA.
- Available in a range of allelic frequencies (Afs) from 0.1% to 5% as purified DNA Mix or encapsulated DNA in synthetic plasma
Seraseq® ctDNA MRD Panel
- An ultra-low allelic frequency ctDNA reference material for development and limit of detection (LoD) validation of NGS-based MRD assays
- Derived from a diseased cell line harboring a high number of somatic variants, its SNP-matched normal cell line, and additional therapeutically relevant biosynthetic DNA variants
- Tumor and normal DNA is blended to four tumor fractions (0.5%, 0.05%, 0.005% and 0%), fragmented and sized to mimic patient ctDNA; all blends are characterized by NGS
Better assays and more thorough validations
- Confidently evaluate your assay's concordance with tissue-based tests using allele frequencies up to 5%
- ctDNA-like size distribution and efficient library incorporation enhance NGS workflow performance and data quality
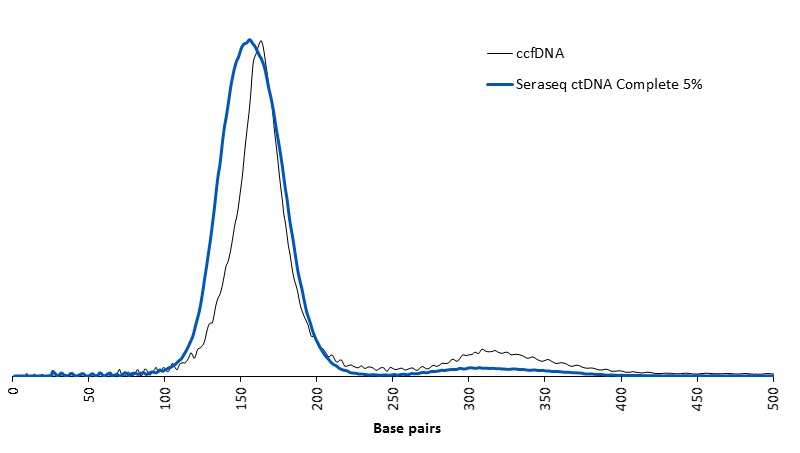 |
Representative DNA fragment size distribution for the Seraseq ctDNA Complete™ Mutation Mix AF5% versus natural circulating cell-free DNA (ccfDNA)
|
Ensure assay sensitivity and accuracy
- Determine your assay’s LOD with orthogonally validated allele frequencies down to 0.1%
- Confidently establish your assay’s ability to detect SNVs, INDELs, CNVs, and SVs with reference materials featuring the broadest coverage of all variant types
- Expert-designed content features up to 40 clinically relevant variants associated with diagnosis, disease progression and resistance monitoring, and therapeutic selection
Gene List
| AKT1 ■ ● |
BRCA1 ● |
ERBB2 ■ ● |
GNA11 ■ |
KIT ■ ● |
NCOA4-RET ■ ● |
PTEN ■ |
| ALK ● |
BRCA2 ● |
ERBB2 CNV ● |
GNAQ ■ |
KRAS ■ ● |
NPM1 ■ |
RET ■ |
| APC ■ |
CD74-R0S1 ● |
FGFR3 ■ |
GNAS ■ |
MET CNV ● |
NRAS ■ ● |
SMAD4 ■ |
| ATM ■ |
CTNNB1 ■ |
FLT3 ■ |
IDH1 ■ |
MPL ■ |
PDGFRA ■ |
TP53 ■ |
| BRAF ■ ● |
EGFR ■ ● |
FOXL2 ■ |
JAK2 ■ |
MYC CNV ● |
PIK3CA ■ ● |
TPR-ALK ■ ● |
ctDNA v2 ■ ctDNA complete ●

