IVD Assay Development
Seraseq® FFPE HRD Negative RM
Details
Resources
Specifications
Frequently Asked Questions
•Derived from tumor and matched-normal human cell lines
•Blended to ~65% tumor content with SNP-matched normal cell lines
•Additional biosynthetic single-nucleotide variants (SNVs) of 4 homologous recombination repair (HRR) genes
•10 µm FFPE curl with DNA yield >100 ng per curl
•Full-process FFPE format reference material validated by NGS and microarray assays with various HRD algorithms (see the Data Sheet for more details)
•Manufactured in GMP-compliant, ISO 13485-certified facilities
Seraseq FFPE HRD (High-Positive, Low-Positive, Negative) Reference Material Package Insert
Seraseq FFPE HRD Negative Reference Material
Technical Spreadsheet for Seraseq HRD Reference Materials
Seraseq FFPE HRD (High-Positive, Low-Positive, Negative) Reference Material
Seraseq FFPE HRD (High-Positive, Low-Positive, Negative) Reference Material Product Sheet
Seraseq FFPE Safety Data Sheet
Seraseq FFPE HRD Negative Reference Material
Seraseq FFPE HRD Negative Reference Material
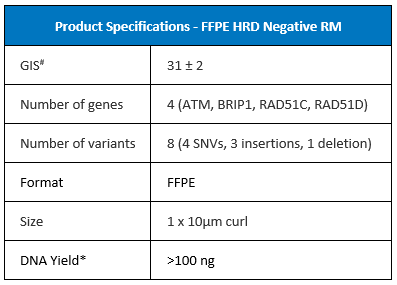
#Genomic Instability Score (GIS) calculated using Illumina TruSight™ Oncology (TSO) 500 HRD RUO assay which calculates a GIS using an algorithm licensed from Myriad Genetics. Illumina TSO 500 HRD is not available in Japan.
*From 1x10 µm FFPE curl using QIAGEN QIAamp FFPE DNA Tissue Kit/Qubit DNA HS quant
Solid Tumor FAQs
Review the common questions we receive from our customers and the responses we provide.


